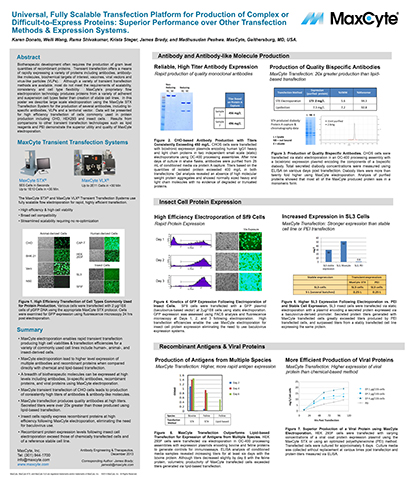
Universal, Fully Scalable Transfection Platform for Production of Complex or Difficult-to-Express Proteins: Superior Performance over Other Transfection Methods & Expression Systems
Biotherapeutic development often requires the production of gram level quantities of recombinant proteins.
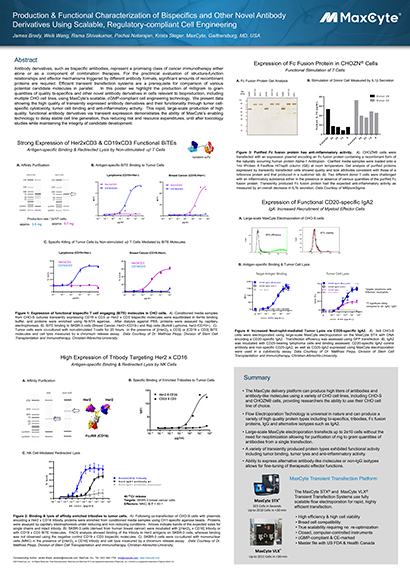
Read MoreProduction & Functional Characterization of Bispecifics and Other Novel Antibody Derivatives Using Scalable, Regulatory-compliant Cell Engineering
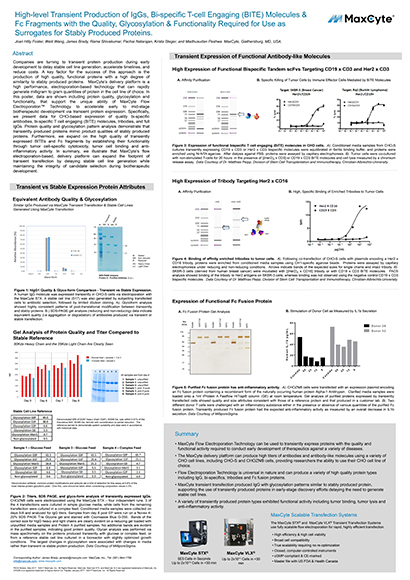
High-level Transient Production of IgGs, Bi-specific T-cell Engaging (BITE) Molecules & Fc Fragments with the Quality, Glycosylation & Functionality Required for Use as Surrogates for Stably Produced Proteins
Companies are turning to transient protein production during early development to delay stable cell line generation, accelerate timelines, and reduce costs. A key factor for the success of this approach is the production of high quality, functional proteins with a high degree of similarity to stably produced proteins.
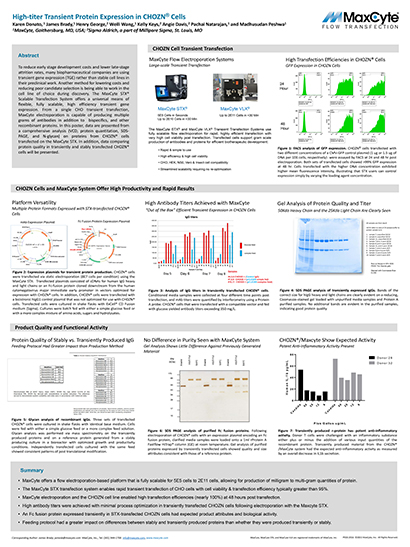
Read MoreHigh-titer Transient Protein Expression in CHOZN®Cells
To reduce early stage development costs and lower late-stage attrition rates, many biopharmaceutical companies are using transient gene expression (TGE) rather than stable cell lines in their preclinical work. Another method for lowering costs and reducing poor candidate selection is being able to work in the cell line of choice during discovery.
Read MoreLentivirus and AAV Production in Suspension and Adherent Cells with MaxCyte Flow Electroporation
Viral Vectors have been a core component of gene therapy since the 1980s. Since that time vector production technologies have not changed a great deal, leaving industry and academia alike to face the same challenges.
Read MoreMaximizing Productivity and Data Relevance with a Scalable, Non-viral Cell Engineering Technology
Protein-based and cellular therapeutics have revolutionized the treatment of cancer and other diseases. However, the need to produce complex biologics with limited resources and shortened timelines has created a demand for technologies that can express multiple therapeutic moieties in a range of cell types and culture processes.
Read MoreOptimization of CHO Transient Gene Expression (TGE) for Multi-Gram Level Antibody Production: Effects of Expression Construct, Post Transfection Cell Density and Feed Conditions
A variety of CHO cell transient transfection methods have been reported including systems based on engineered CHO cells, unique expression systems and specialized transfection reagents, but they each have varying levels of reproducibility, scalability, and cost effectiveness and generally produce antibody titers from 10 – 100mg/L (1-5).
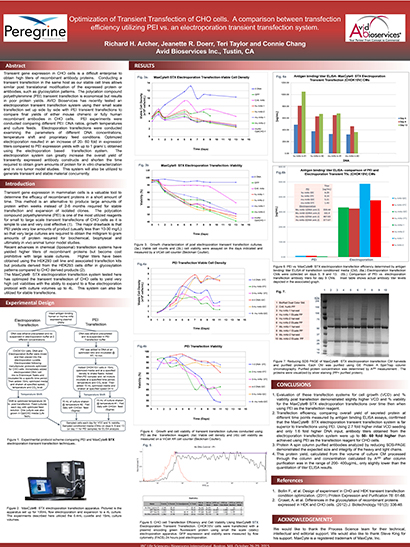
Read MoreOptimization of Transient Transfection of CHO cells. A comparison between transfection efficiency utilizing PEI vs. an electroporation transient transfection system
Transient gene expression in CHO cells is a difficult enterprise to obtain high titers of recombinant antibody proteins. Conducting a transient transfection in the same host as our stable cell lines allows similar post translational modification of the expressed protein or antibodies, such as glycosylation patterns.
Read MoreOvercoming Transient & Stable Productivity Challenges to Efficiently Scale Protein Production from Early Discovery Through Clinical Translation.
Navigating protein production from early-stage discovery research through therapeutic manufacturing is faced with a variety of challenges such as, low-titer transient productivity, lack of scalability, required re-validation upon switching expression systems, and the extended time for stable cell generation.
Read MoreProtein Production in Insect Cells: Flow Electroporation, a Superior Alternative to Baculovirus Expression
Insect cells are used for the industrial manufacturing of many products that are currently in clinical trials or already available on the market for veterinary and human applications.
Read More