Rapid and Stable Protein Production with Consistent Quality to Accelerate Biotherapeutic Development Regulatory Sciences for Biologics & Vaccines
Rapid recombinant protein production is one of the key elements for drug development success.
Read MoreGene Editing: Paving the Way for Accelerated Clinical Development of Adoptive Cell Immunotherapies
Precision genome engineering requires technologies that allow efficient and reproducible delivery of DNA, mRNA and RNP-based reagents into a range of primary cells and stem cells. In addition, clinical gene editing requires a transfection platform that is GMP-compliant and scalable to accommodate billions of cells in a single transfection.
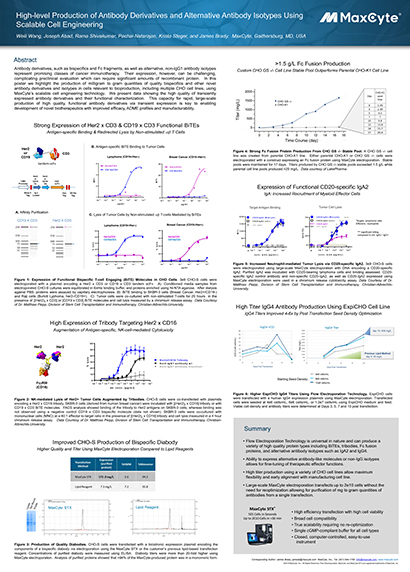
Read MoreHigh-level Production of Antibody Derivatives and Alternative Antibody Isotypes Using Scalable Cell Engineering
Antibody derivatives, such as bispecifics and Fc fragments, as well as alternative, non-IgG1 antibody isotypes represent promising classes of cancer immunotherapy. Their expression, however, can be challenging, complicating preclinical evaluation which can require significant amounts of recombinant protein.
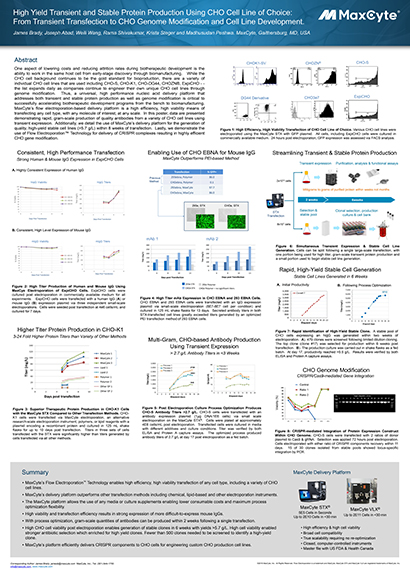
Read MoreHigh Yield Transient and Stable Protein Production Using CHO Cell Line of Choice: From Transient Transfection to CHO Genome Modification and Cell Line Development
One aspect of lowering costs and reducing attrition rates during biotherapeutic development is the ability to work in the same host cell from early-stage discovery through biomanufacturing.
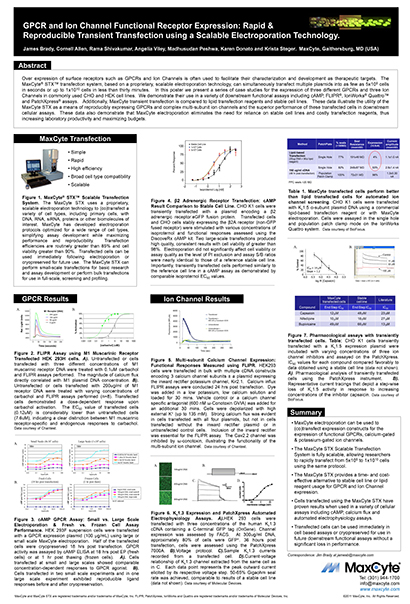
Read MoreGPCR and Ion Channel Functional Receptor Expression: Rapid & Reproducible Transient Transfection using a Scalable Electroporation Technology
Over expression of surface receptors such as GPCRs and Ion Channels is often used to facilitate their characterization and development as therapeutic targets.
Read MoreGMP-compliant Non-viral CRISPR-mediated Process Correcting the Sickle Cell Disease Mutation in SCD Patient CD34+ Cells Achieves 60% Wild Type Adult Hemoglobin Expression in Differentiated Erythrocytes.
The c.20A>T mutation of -globin gene causes sickle cell disease (SCD). Allogeneic hematopoietic stem cell (HSC) transplantation can cure SCD, but most lack a suitable donor. Ex vivo gene therapy strategies, including lentiviral mediated gene transfer or endonuclease mediated BCL11a knockdown allowing fetal hemoglobin (Hb) induction, are currently under evaluation.
Read MoreGetting the Most from Your Manufacturing Cell Line: Reducing the Time and Cost of Progressing to the Clinic Using MaxCyte’s Delivery Platform.
The combination of choosing the correct host cell line and the right delivery platform both have significant impacts on the time and cost of progressing to the clinic.
Read MoreGeneration of Assay-ready Cells and Functional Cell-based Assays for Ion Channels and Transporters Using MaxCyte’s Universal Cell Engineering Platform
Cell-based assays using mammalian cells to express receptor targets are widely used to increase our understanding of the molecular mechanisms of human disease and to develop novel therapeutics.
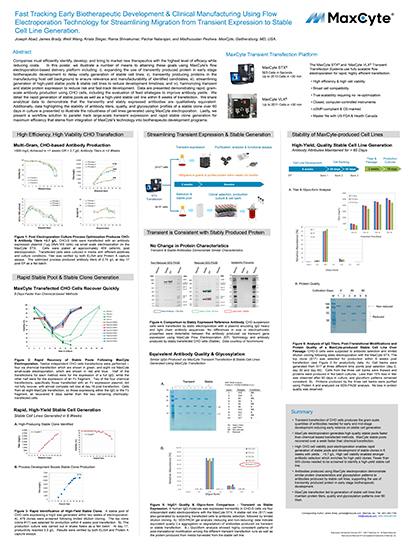
Read MoreFast Tracking Early Biotherapeutic Development & Clinical Manufacturing Using Flow Electroporation Technology for Streamlining Migration from Transient Expression to Stable Cell Line Generation
Companies must efficiently identify, develop, and bring to market new therapeutics with the highest level of efficacy while reducing costs.
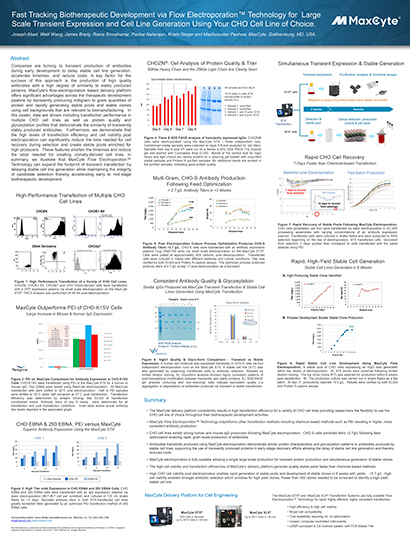
Read MoreFast Tracking Biotherapeutic Development via Flow Electroporation™ Technology for Large Scale Transient Expression and Cell Line Generation Using Your CHO Cell Line of Choice
Companies are turning to transient production of antibodies during early development to delay stable cell line generation, accelerate timelines, and reduce costs. A key factor for the success of this approach is the production of high quality antibodies with a high degree of similarity to stably produced proteins.
Read More