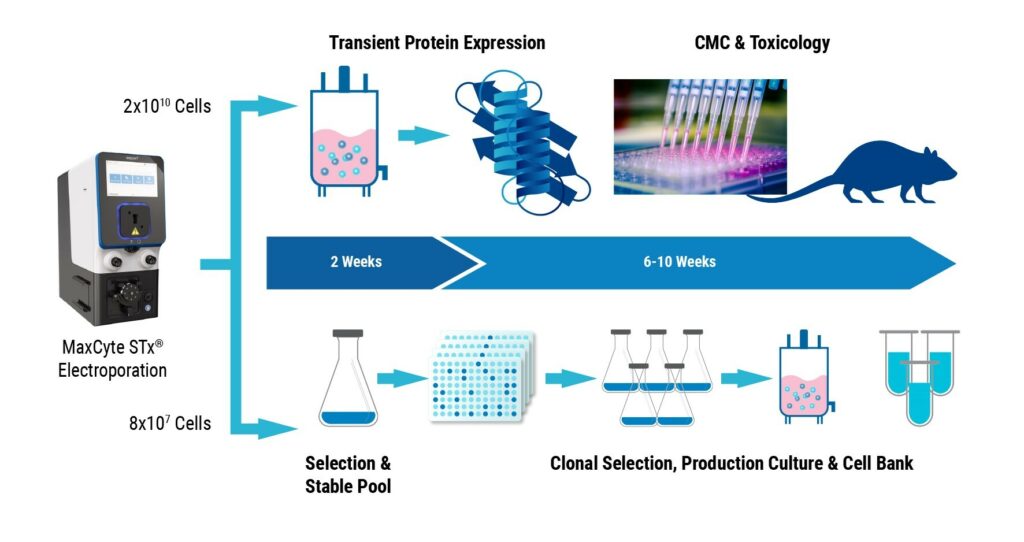
Developing Assays for Screening GPCRs with Transiently Transfected Cells Using the MaxCyte® STX™ System
G protein coupled receptors (GPCRs) represent targets for at least one third of drugs that are cur rently marketed, and they are of central importance to ongoing drug discovery efforts throughout the pharmaceutical industry.
Read MoreDeveloping a Luciferase-Based NF-κB Reporter Assay with Transiently Transfected HEK 293 Cells
Firefly luciferase is widely used as a reporter mole cule in cell based assays because of its sensitivity, dynamic range and ease of detection.
Read MoreAccelerating Discovery and Development: Reducing Risk with High Performance Transfection of the Right Cell for the Right Application
To remain competitive in today’s world, pharmaceutical, biopharmaceutical, and biotechnology companies must quickly identify, develop, and bring candidates to market with high efficiency at the lowest cost.
Read MoreLarge-Scale Engineering of Extracellular Nanoparticles for Genome Editing with CRISPR-Cas9 Ribonucleoproteins
Despite advances in human genetics, patients with rare diseases such as Duchenne muscular dystrophy (DMD) still have limited therapeutic options and a short life expectancy.
Read MoreMaxCyte® Enables Development of New, Rapid, Sensitive SARS-CoV-2 Antibody Test
MaxCyte Flow Electroporation® technology helped researchers develop and implement a new diagnostic test for SARS-CoV-2 infection that is quick, sensitive and easily adaptable as novel pathogenic strains emerge.
Read MoreMaxCyte® Enables a cGMP-Compatible Manufacturing Process for Highly Efficient T Cell Engineering
Excitement over recent breakthroughs in autologous cellular therapies has been tempered by the expense of manufacturing with viral gene delivery and concerns over random integration and the safety of viral vectors.
Read MoreMaxCyte® Enables Multiplexed NK Cell Engineering
As the immune system’s first responders, natural killer (NK) cells are essential for the rapid detection and elimination of cancer. Their ability to kill cancer cells without prior sensitization makes them an ideal immunotherapy candidate.
Read MoreAdvancing a CCR5 Gene Editing Therapy to Clinical Trial: Overcoming Viral Vector Toxicity
HIV infection is currently managed by lifelong antiretroviral therapy (ART), a modality that is associated with chronic toxicity, challenging patient compliance, and comes at a significant cost over a patient’s lifetime.
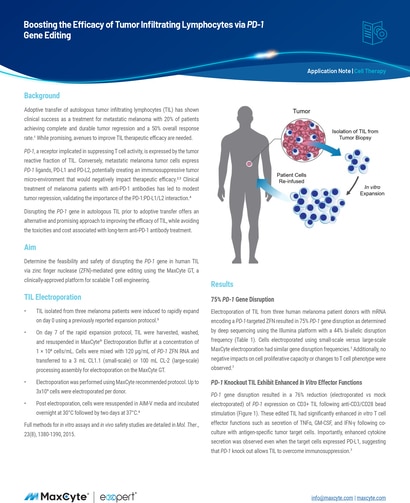
Read MoreBoosting the Efficacy of Tumor Infiltrating Lymphocytes via PD-1 Gene Editing
Adoptive transfer of autologous tumor infiltrating lymphocytes (TIL) has shown clinical success as a treatment for metastatic melanoma with 20% of patients achieving complete and durable tumor regression and a 50% overall response rate.1 While promising, avenues to improve TIL therapeutic efficacy are needed.
Read MoreEnhancing HIV-1 Vaccine Efficacy and Manufacturing Through CHO Cell CRISPR Engineering
The RV144 HIV vaccine trial, the first trial to demonstrate that vaccination could confer HIV protection, reported a modest 31% efficacy. gp120, a major component of the RV144 trial, was produced in Chinese Hamster Ovary (CHO) cells and lacked N-linked glycosylation sites critical for binding of anti-HIV broadly neutralizing antibodies (bN-mAbs).
Read More