Large-Scale Cell Concentration and Buffer Exchange Prepares CHO Cells for Electroporation
Recombinant protein production is an essential step in the development of many novel, life-saving therapies and diagnostics. Stable cell line development enables consistent protein yields and product quality, but the process is lengthy, costly and time-consuming.
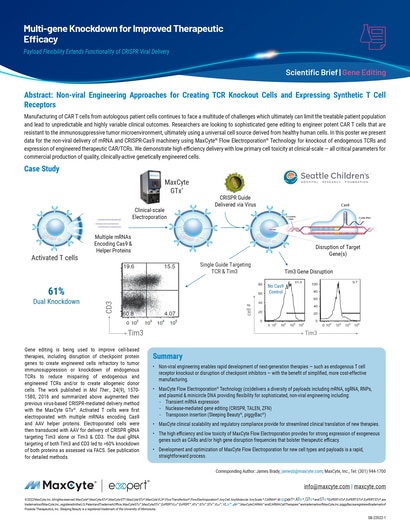
Read MoreMulti-gene Knockdown for Improved Therapeutic Efficacy
Manufacturing of CAR T cells from autologous patient cells continues to face a multitude of challenges which ultimately can limit the treatable patient population and lead to unpredictable and highly variable clinical outcomes.
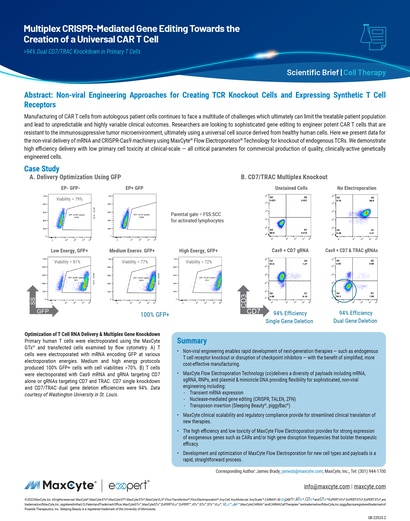
Read MoreMultiplex CRISPR-mediated Gene Editing Towards the Creation of a Universal CAR T Cell
Manufacturing of CAR T cells from autologous patient cells continues to face a multitude of challenges which ultimately can limit the treatable patient population and lead to unpredictable and highly variable clinical outcomes.
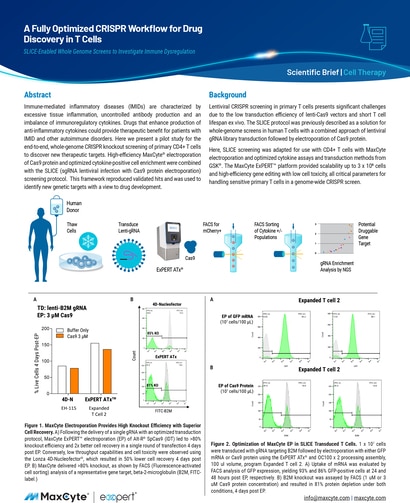
Read MoreA Fully Optimized CRISPR Workflow for Drug Discovery in T Cells
Immune-mediated inflammatory diseases (IMIDs) are characterized by excessive tissue inflammation, uncontrolled antibody production and an imbalance of immunoregulatory cytokines.
Read More